With the rise of energy costs, membrane technology for separating gases plays a significant role in reducing the environmental impact and costs of industrial processes. Polymer-based gas separation membranes offer advantages over conventional technologies (cryogenic distillation, amine absorption, etc.) including, low capital and operating costs, minimum energy requirements, ease of operation, and environmental friendliness. However, gas separation in polymers have reached a seemingly reached the trade-off between permeability and selectivity, as plotted on the 2008 Robeson plot. Inorganic membranes, on the other hand, display excellent permselectivity properties but incur high fabrication costs and lack membrane mechanical stability. Mixed-matrix membranes (MMMs) comprising thermally stable polymers and inorganic additives for target gases combines the polymers' mechanical properties with the additives' permselectivity properties. Our group focuses on MMMs consisting of zeolitic imidazolate framework (ZIF) materials in various polymer matrices, including polybenzimidazole (PBI), 6FDA-based polyimides, and polymers of intrinsic microporosity-1 (PIM-1) for industrially relevant gas separations. ZIFs belong to a subclass of highly porous metal organic frameworks (MOFs) having structures similar to zeolites. The presence of organic linkers in ZIFs results in a better affinity between the polymer and ZIF, compared to a nonfunctionalized zeolite.


_________________________________________________________________________
Polymer Light-Emitting Diodes (PLEDs)
Conjugated polymers have been extensively studied as electroluminescent materials for application in polymer light-emitting diodes (PLEDs), due to their low cost, simple solution processability, and convenience in fine-tuning the emission colors. The commercially available small molecule OLEDs are now found in several small-display applications, such as mobile phones and tablets, because they offer several desirable attributes, such as high resolution and high contrast images, with fast response times and wide viewing angles. These advantages indicate that the PLED technology is one of the most promising candidates for next-generation large area flat-panel displays. The efficiency of PLED devices depends on a number of factors, including: the number of charges injected into the polymer film from each electrode, the mobility of these charges through the polymer film, and finally the charge-balance ratio or recombination efficiency of the holes and electrons within the polymer film. Figure below displays a typical a single layer PLED device.
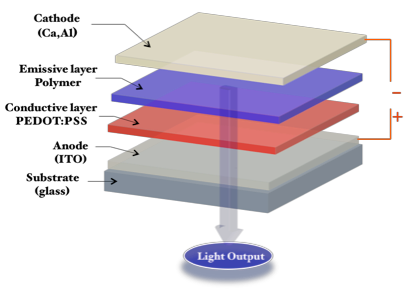
Schematic representation of single-layer PLED device.
Our group focuses on the synthesis and characterization of alkylsulfone -containing poly(phenylene vinylenes) and poly(fluorenevinylenes) for polymeric light emitting devices (PLEDs). The attractive combination of excellent optical and electronic properties of the conjugated polymers has encouraged our synthesis of a novel n-type symmetrical alkylsulfonyl-substituted poly(phenylenethynylene) derivative, poly[2,5-bis-(2'-ethylhexylsulfonyl)-1,4 phenylene)ethynylene] (SO2EH-PPE). This polymer is anticipated to display high solution state quantum yields of fluorescence and typically have wider band gaps than PPVs (potential blue light emitter).

_________________________________________________________________________
Although inorganic-based solar cells harvest solar energy efficiently, polymer-based solar cells (PSCs) have recently attracted much attention due to their potential for low-cost, flexible, and lightweight solar energy harvesting devices. In particular, bulk heterojunction solar cells (BHSCs), comprising physical composites of conjugated polymers and fullerene derivatives, are being intensely studied to achieve higher efficiencies in PSCs. Conducting polymers in these devices play the role of the electron donor while fullerene derivatives act as the electron acceptor. One of the common polymers used in PSCs are the 3-alkyl-substituted polythiophenes, with efficiencies of poly (3-hexylthiophene) and (6,6)-phenyl C61-butyric acid methyl ester (PCBM) composites routinely reaching up to 4-5%.

Our research group focuses on the design and synthesis of new low band-gap polymers for organic solar cells. Among various materials, benzo [1,2-b:4,5-b']dithiophene (BDT) polymers have attracted our interest because of its exceptional performance as a common unit in PSCs with up to ~8 % efficiencies, which represents one of the highest efficiencies reported in the area of PSCs.

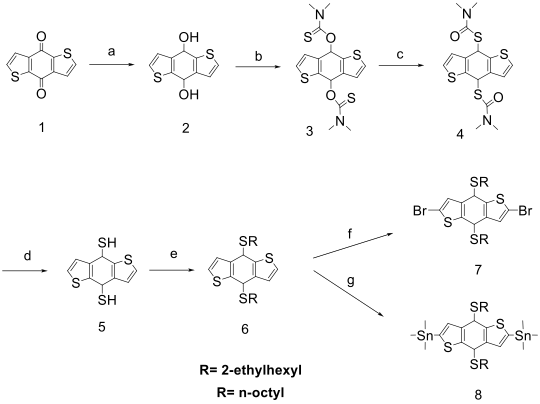
Synthetic routes to precursor monomers

Synthetic routes for semiconducting polymers
_________________________________________________________________________
Supercapacitors, also known as electrochemical double layer capacitors (ECs) or ultracapacitors, are energy devices that store energy by the electric field present between a charged electrode and the counterions of the electrolyte. They possess relatively high energy density in the order of hundreds of times greater compared to conventional capacitors. Compared to conventional batteries or fuel cells, EDLCs also have a much higher power density.

Among various carbon materials, such as particles, papers, or nanotubes, carbon nanofibers are good candidates for electrode materials since they provide high surface area and porosity. These desirable properties can be attributed to their small fiber diameters and an entangled and interconnected structure. Carbon nanofibers are generally prepared by electrospinning of carbon precursor polymers and thermal treatment, which is called carbonization or graphitization.
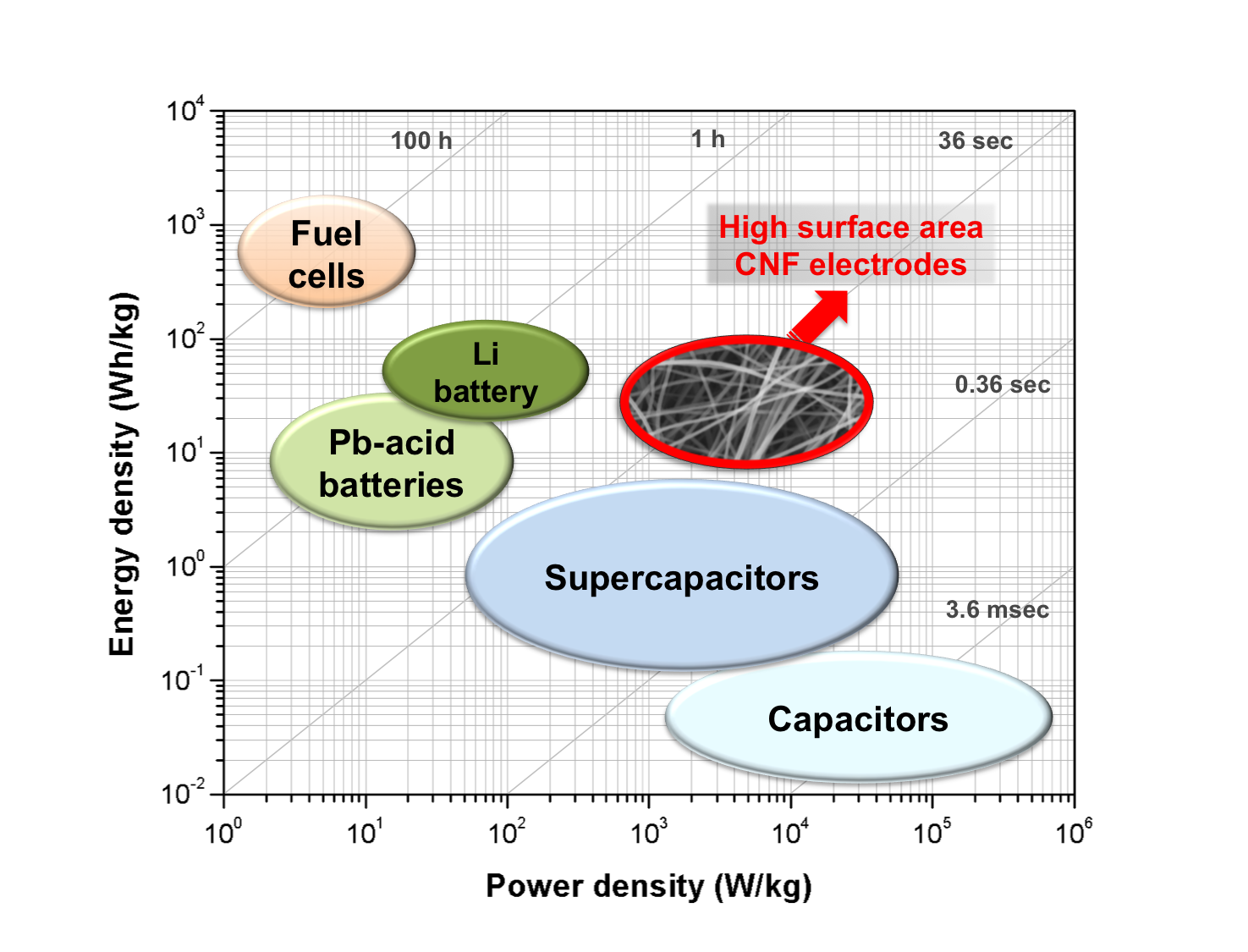
Our research focuses on the preparation of carbon nanofibers derived from new polymer precursors such as blends or copolymers. Specific interactions between polymer precursors determine the resultant carbon structure, which is crucial for supercapacitor performance. We have also investigated several strategies to increase capacitance, energy and power density, including increasing the porosity and conductivity of the carbon-based electrodes, and the use of high voltage electrolytes such as ionic liquids. Increasing the surface area in carbon fibers can be achieved by the inclusion of pore-forming agents, such as volatile salts, polymer sacrificial templates, or polymers with inherent porosity upon carbonization.
_________________________________________________________________________
Fuel cells are electrochemical devices that convert the chemical energy of the fuel and oxidant directly into electricity and heat. Unlike a regular battery, a fuel cell can essentially function as a power source as long as there is supply of fuel (H2 or methanol). Our group, in collaboration with the UTD Fuel Cell Team, is focusing on the preparation and characterization of polymer-based materials for proton exchange membrane (PEM) fuel cells. We have worked with organic-inorganic membrane hybrids (see Publications), and more recently, on perfluorocyclobutyl (PFCB)-based polymer blends.
Commercialization of PEM fuel cells has been hampered due to the limitations in the widely used polymer, Nafion. This perfluorinated block copolymer has limited performance at temperatures greater than 100 oC and low relative humidity conditions (RH less than or equal to 50 oC), making it unsuitable for transportation applications. Furthermore, Nafion undergoes structural changes with dehydration, making it unpredictable under the desired fuel cell operating conditions. In addition, membrane durability and cost are also issues to overcome. There has been a number of strategies to improve or replace Nafion, and one of the most effective is to mimic Nafion's nanophase-separated morphology through the use of block copolymers of hydrophobic and hydrophilic blocks.
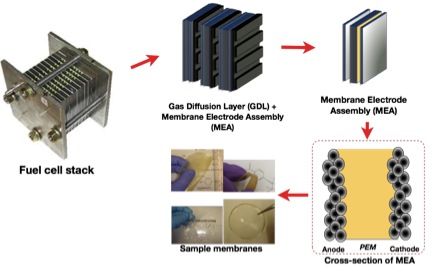
Chemically, mechanically and thermally stable PFCB polymers are ideal to use for PEMFC applications. These polymers can be synthesized via a facile and condensate-free thermal polymerization with high molecular weights from commercially available and cheap starting materials. However, in the preparation of PFCB-based block copolymers, the polymerization of the hydrophilic and hydrophobic oligomers require different polymerization rates, making the control of length of block segments and molecular weights difficult. An easier route to obtaining a block copolymer morphology, thereby mimicking Nafion's morphology, is through blending PFCB-based hydrophilic and hydrophobic polymers.
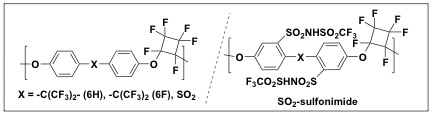
We have synthesized PFCB polymers with (-SO2-, -C(CF3)2-, -CO-) and without (biphenyl, BP) bridging groups, and sulfonated SO2-PFCB and BP-PFCB with sulfonimide and sulfonic acid moieties, respectively. Blends of various compositions and ratios are currently investigated and characterized.